The new science of the microbiome suggests that we may have been thinking about dieting and body size all wrong.

Imagine a very strange experiment.
We take two people, and we feed them the exact same meal—making sure they eat every bite. A day or two later, when that meal comes out the other end of them as poop, we collect it. (Well…we have them collect it.)
We dry it out, and light it on fire in a device called a “calorimeter,” which enables us to measure how much heat comes off when something burns—that is, how much energy is in it: how many calories.
In both people, it was the same number going in. But will it be the same number coming out?
The answer is no.
Strange as it might sound, an experiment like this has recently been done, and it’s just one of the thousands of studies from the past few years changing the way we think about dieting and nutrition by looking at these topics through the lens of the microbiome: the trillions of bacteria that live in and on your body, primarily in your gut.1 The variability of the microbiome from person to person means that the same food can have wildly different effects on the bodies of two different people. While more research is needed to fully uncover these differences in humans, animal experiments have shown that sterile mice (those without a microbiome) who have a slower aerobic metabolism can eat more—but still weigh less—than ordinary mice (those with a microbiome) who consume the same diet.2
Another layer of complexity? Even though the microbiome is generally stable, once it reaches the “steady state” of adulthood, the relative amounts of different bacteria can vary day to day within a person, depending on variables like dietary choices.3 So even if that person consumes the same meal twice, one week apart, that meal might have different effects on their body, depending on what they ate in the intervening days. But calories are only the start. New research is implicating the microbiome in determining everything from basal metabolic rate (the amount of calories you burn just by existing) to satiety signaling (how full you feel after a meal).
As you can imagine, these nuances mean that dieting is far from a one-size-fits-all (sorry) endeavor. They may also provide insight into why the “perfect” diet feels so elusive, why body composition varies among people who consume relatively similar foods, and why dieting remains an enduring scientific and cultural fascination (and frustration). Here, we’ll dig into a few ways that the new science of the microbiome is changing our understanding of our relationship with food, and how this science can help inform how we think about dieting and weight.

Science with us—nerdy reads for your inbox biome1
1By signing up, you consent to receive Seed emails.
Thinking Beyond “Fast” and “Slow” Metabolism
Possibly the most worn-down piece of diet advice that’s still trotted out is: “calories in, calories out.” It’s simple physics, the thinking goes. Burn more calories than you take in as food, and you will inevitably lose weight. And while it’s true that you can’t beat thermodynamics, the body is complicated. Consider the fact that two people can burn vastly different numbers of calories even while sitting perfectly still.
This concept, known as your basal metabolic rate, is what people are typically referring to when they say they have a “fast” or “slow” metabolism. But there’s a falsehood hidden in this language: the notion that the speed of your metabolism is something inborn, or beyond our control. For instance, most tests for basal metabolic rate work by measuring how much oxygen is converted to carbon dioxide (CO2) in a person’s breath, over a set period of time. This works because the body’s metabolism is aerobic: Like a candle flame, it turns fuel and oxygen into energy and CO2. If you measure the amount of oxygen consumed by a candle, you can tell how much fuel it’s burning, and how much energy it’s producing. The same is true, in principle, for humans.
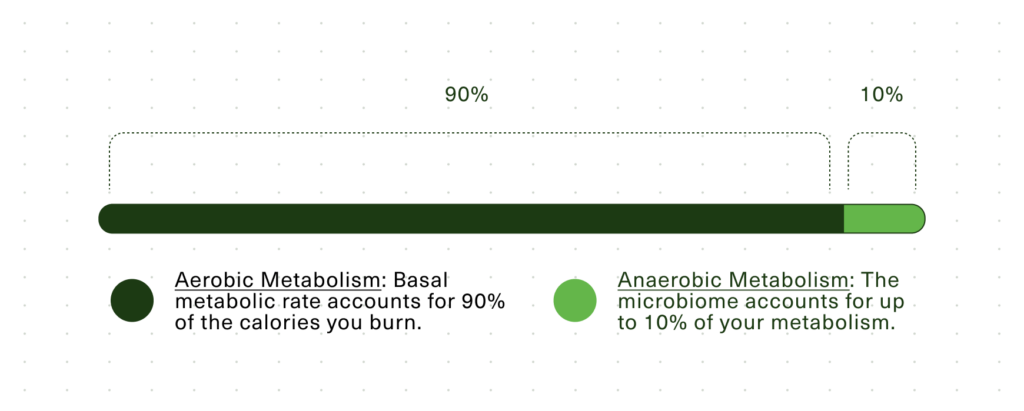
But humans aren’t candles, and if you actually measure the heat output of a human, and compare it to the amount of oxygen they use, you’ll find a mismatch: Aerobic respiration can only account for about 90% of most people’s heat output.4
That mysterious remaining 10% leads us back to the microbiome, which primarily uses anaerobic (not aerobic) metabolism to turn calories from food into energy without oxygen getting involved. But not everybody’s microbiome is the same. Research has shown that this aerobic/anaerobic discrepancy is simply missing in some people with obesity.4 Their aerobic metabolism is responsible for 100% of their heat output. In other words, they’re not getting an anaerobic assist from their microbiome.
When you think about it, 10% of your daily calories is a lot. A 2018 report from the UK Health Security Agency estimates that “overweight and obese boys consume anywhere between 140–500 calories too many each day, depending on their age. For overweight and obese girls, it’s 160–290.” 5 Based on that, a difference of 10% is more than enough to have a major impact on a person’s ability to lose weight.
Direct interventional research in humans bears this out as well. In a 2021 study, researchers found that people placed on a calorie-restricted diet (at a daily “deficit” of 500 calories) all lost weight, but the amount they lost depended heavily on the composition of their microbiome at baseline.6 In fact, they were able to reliably model whether or not a person would lose more versus less than 5% of their body weight after 3.5 months of calorie-restriction, based solely on the composition of their microbiome when they started.
It’s important to mention here that the microbiome’s impact on metabolism isn’t always beneficial. For some, it seems, the microbiome simply makes losing weight harder—but why?
Inflammation is one clear driver: Many kinds of bacteria are covered in a highly pro-inflammatory molecule called lipopolysaccharide, or LPS for short. When LPS gets into the bloodstream, it can reprogram the body to accumulate more visceral fat, and can cause insulin resistance—one of the key hallmarks of diabetes. In 2013, researchers found that “germ-free” mice (that is, mice with no microbiome whatsoever) don’t become obese even on a high-fat diet. However, introducing a strain of Enterobacter isolated from an obese human caused the mice on the high-fat diet (but not a regular diet) to rapidly gain weight.7
Enterobacter isn’t the only culprit here, though. Even bacteria without LPS can drive issues with metabolism and weight, as evidenced by the results of that calorie-restriction study: In people who lost at least 5% of their body weight, the low-calorie diet reduced the abundance of a bacterial genus called Enterococcus (another opportunistic pathogen which can trigger an inflammatory immune response) in their guts to about one one-millionth of its previous levels.
While this study singled out Enterococcus, this is likely just one of many culprits that can drive inflammation and weight gain. Only 30% of the study group lost more than 5% of their body weight during the calorie-restriction period, and these were the people who saw a significant reduction in Enterococcus levels. In the other 70%, who didn’t lose as much weight, there was no significant change in Enterococcus. There’s clearly a lot left to learn here, but this is still exciting: It suggests that for some people, the secret to weight loss is actually in defeating the inflammatory components of their microbiome—and that calorie restriction is just a means to this end.
Fat, Fiber, and Satiety
“Eat fewer calories” is simple advice, but the distinction between “simple” and “easy” makes all the difference in the world. For many people, a daily deficit of 500 calories is a recipe for perpetual “hangriness” that can make ordinary activities seem impossible. But some people seem to be more susceptible to this sensation than others. As we learn more about the body, it’s becoming clear that the major difference between people who find it easy to lose weight and those who find it difficult isn’t one of willpower, but a biological difference in satiety: how full you feel after a certain amount of food, and how long it keeps you feeling full.
Satiety is regulated by a number of biological factors in the body. You might already be familiar with the most common example of satiety regulation: the interplay between insulin and blood sugar. Carbohydrates from food are absorbed into the bloodstream as glucose, and this diminishes feelings of hunger while also stimulating the release of insulin from the pancreas. Insulin spurs your muscles and other cells to absorb that glucose from the bloodstream, enabling it to be turned into energy. When the body has burned through that fuel, hunger starts to return. But rapid spikes in blood sugar can cause the body to “overshoot” on insulin production, which is part of why sugary snacks often leave us hungry again a little while later.
While that is the classic example of satiety regulation, a new target for controlling appetite has been getting a lot of attention in recent years: a molecule called glucagon-like peptide 1, or GLP-1.
GLP-1 is a peptide (a small protein) that your gut produces to signal to other parts of your body. When you eat, it’s released from cells in your intestines, and lands on receptors that are found in your pancreas, as well as on the vagus nerve—the most direct line of communication from your gut to your brain. It’s a bit of an oversimplification, but you can think of the GLP-1 receptor as the “full” button in your brain, and GLP-1 itself as the finger that presses that button. This is how the popular new drug Ozempic works, encouraging satiety by artificially stimulating the GLP-1 receptor.
But what’s going on in some people’s bodies such that they need extra help from Ozempic in order to feel full? One clue comes from how GLP-1 itself is regulated. The release of GLP-1 isn’t triggered by the physical sensation of food in the intestines; it’s triggered by chemical signaling, when receptors on the intestinal cells are activated by compounds in the food we eat—and by signals produced when gut bacteria transform those compounds.
Fat-rich foods generally stimulate GLP-1 release better than carbohydrates, which is part of why the high-fat, ultra-low-carb “keto” diet (originally developed for the treatment of epilepsy) has found adherents in the world of weight loss. In a 2015 study, which looked at 12 healthy white men, researchers found that simply feeding these men olive oil (with a little shredded carrot) was enough to induce a major spike in GLP-1 levels, leading to the sensations of satiety similar to the ones Ozempic creates pharmaceutically. While this study is admittedly small and not generalizable—for one, it didn’t include any women—it suggests that there’s a relationship between olive oil and the release of GLP-1, at least in some people.8
But it may not be the olive oil by itself that’s responsible: The gut bacterial genus Gemella produces a molecule called oleoyl serinol, which is known to stimulate the release of GLP-1 by intestinal cells—and the microbe relies on dietary oleic acid, which is found at high levels in olive oil, to do so.9 Fats are generally absorbed early in the digestive process (in the small intestine) before they ever reach the colon where most of your bacteria live, but Gemella is one type of microbe that lives in the mucus lining of the small intestine, giving it a chance to turn these dietary fats into a feeling of fullness. Gemella isn’t unique in this respect, either. A 2021 study found that a human gut bacterium called Holdemanella biformis could also enhance GLP-1 release in mice.10
Fat isn’t the only chemical signal involved in GLP-1 release, though: A number of animal studies have found short-chain fatty acids (SCFAs for short) to be major regulators of the peptide.11 Because these molecules are produced mainly by gut bacteria when you eat fiber, this suggests that fiber is also a key player in helping us feel full. And because SCFAs are produced in a slow and steady way, starting several hours after a meal, fiber may help keep you full in a way that other foods don’t.
However, many of these studies were done in mice or rats, so it’s always important to take findings like that with a grain of salt. In the same study discussed earlier, where researchers found that olive oil could raise levels of GLP-1 in humans, it was also shown that artificially elevating levels of the key SCFA butyrate didn’t exert the same effect on “healthy white men” (in this case, those who didn’t have diabetes or prediabetes and whose BMIs were in what the CDC considers the “healthy range”).12 This might be due to differences in the genetics or microbiome between rodents and humans: Their diets and digestive tracts are pretty different from ours, and so are their behaviors (most humans don’t eat their own poop, for one thing).
The study was conducted in “healthy” individuals, though, so another possibility is that people who aren’t overweight and who eat a healthy, balanced diet are more likely to be consuming enough fiber, and therefore have sufficient levels of GLP-1. So any boost of SCFAs won’t change the fact that their GLP-1 receptors are already getting what they need to function properly. That said, someone whose microbiome isn’t producing enough SCFAs might still benefit from a microbiome-targeted intervention like a fiber-focused diet if they’re indeed not consuming enough fiber.13
Although this new science is revolutionizing our understanding of how the body works, much of what it teaches us simply provides new reasons to follow “common sense” nutritional advice: Whole grains, fresh fruits, and vegetables provide unique kinds of fiber which help the gut bacteria that are essential for health to thrive, while processed foods that are high in salt and sugar suppress “good” bacteria and feed inflammatory microbes that can contribute to disease.14 But because the microbiome can be sculpted over time, the effects of our dietary choices can be cumulative, meaning that every healthy choice—however difficult it might be—makes the next one easier.
All in all, as new science and new technology change the way we think about weight, the microbiome seems to represent a beacon of hope. On both sides of the “calories in, calories out” equation, we’re finally coming to grips as a society with the fact that biology, rather than willpower, is the real factor underlying some people’s struggle with weight. But just because something is biological doesn’t mean it’s inherent, or immutable: The foods we choose have both short- and long-term impacts on the microbiome, and that in turn can have a huge impact on both the physical and psychological aspects of our relationship with food. Hopefully, even as publication after publication declares this to be the “Age of Ozempic,” we can leave the conversations about dieting and moral fiber in the past, and focus on the real stuff instead.
Citations
- Boekhorst, J., Venlet, N. V., Procházková, N., Hansen, M., Lieberoth, C. B., Bahl, M. I., Lauritzen, L., Pedersen, O., Licht, T. R., Kleerebezem, M., & Roager, H. M. (2022). Stool energy density is positively correlated to intestinal transit time and related to microbial enterotypes. Microbiome, 10(1). https://doi.org/10.1186/s40168-022-01418-5
- Bäckhed, F., Ding, H., Wang, T., Hooper, L. V., Koh, G. Y., Nagy, A., Semenkovich, C. F., & Gordon, J. I. (2004). The gut microbiota as an environmental factor that regulates fat storage. Proceedings of the National Academy of Sciences of the United States of America, 101(44), 15718–15723. https://doi.org/10.1073/pnas.0407076101
- Valdes, A. M., Walter, J., Segal, E., & Spector, T. D. (2018). Role of the gut microbiota in nutrition and health. BMJ, k2179. https://doi.org/10.1136/bmj.k2179
- Riedl, R. A., Atkinson, S. N., Burnett, C. M. L., Grobe, J. L., & Kirby, J. R. (2017). The Gut Microbiome, Energy Homeostasis, and Implications for Hypertension. Current hypertension reports, 19(4), 27. https://doi.org/10.1007/s11906-017-0721-6
- Tedstone, A. (2018, March 6). Why we are working to reduce calorie intake. UK Health Security Agency. https://ukhsa.blog.gov.uk/2018/03/06/why-we-are-working-to-reduce-calorie-intake/
- Dong, T. S., Luu, K., Lagishetty, V., Sedighian, F., Woo, S. O., Dreskin, B. W., Katzka, W., Chang, C., Zhou, Y., Arias-Jayo, N., Yang, J., Ahdoot, A. I., Ye, J., Li, Z., Pisegna, J. R., & Jacobs, J. P. (2021). The intestinal microbiome predicts weight loss on a Calorie-Restricted diet and is associated with improved hepatic steatosis. Frontiers in Nutrition, 8. https://doi.org/10.3389/fnut.2021.718661
- Fei, N., & Zhao, L. (2013). An opportunistic pathogen isolated from the gut of an obese human causes obesity in germfree mice. The ISME journal, 7(4), 880–884. https://doi.org/10.1038/ismej.2012.153
- Mandøe, M. J., Hansen, K. B., Hartmann, B., Rehfeld, J. F., Holst, J. J., & Hansen, H. S. (2015). The 2-monoacylglycerol moiety of dietary fat appears to be responsible for the fat-induced release of GLP-1 in humans. The American journal of clinical nutrition, 102(3), 548–555. https://doi.org/10.3945/ajcn.115.106799
- Olson, C. A., Vuong, H. E., Yano, J. M., Liang, Q. Y., Nusbaum, D. J., & Hsiao, E. Y. (2018). The Gut Microbiota Mediates the Anti-Seizure Effects of the Ketogenic Diet. Cell, 173(7), 1728–1741.e13. https://doi.org/10.1016/j.cell.2018.04.027
- Romaní-Pérez, M., López-Almela, I., Bullich-Vilarrubias, C., Rueda-Ruzafa, L., Del Pulgar, E. M. G., Benítez-Páez, A., Liebisch, G., Lamas, J. A., & Sanz, Y. (2021). Holdemanella biformis improves glucose tolerance and regulates GLP‐1 signaling in obese mice. The FASEB Journal, 35(7). https://doi.org/10.1096/fj.202100126r
- Tolhurst, G., Heffron, H., Lam, Y. S., Parker, H. E., Habib, A. M., Diakogiannaki, E., Cameron, J., Grosse, J., Reimann, F., & Gribble, F. M. (2012). Short-chain fatty acids stimulate glucagon-like peptide-1 secretion via the G-protein-coupled receptor FFAR2. Diabetes, 61(2), 364–371. https://doi.org/10.2337/db11-1019
- Assessing your weight. (2023, June 9). Centers for Disease Control and Prevention. https://www.cdc.gov/healthyweight/assessing/index.html
- O’Connor, A. (2023, June 28). Are all calories created equal? Your gut microbes don’t think so. Washington Post. https://www.washingtonpost.com/wellness/2023/06/13/weight-loss-calories-fiber-microbiome/#
- Wilck, N., Matus, M. G., Kearney, S. M., Olesen, S. W., Forslund, K., Bartolomaeus, H., Haase, S., Mähler, A., Balogh, A., Markó, L., Vvedenskaya, O., Kleiner, F. H., Tsvetkov, D., Klug, L., Costea, P. I., Sunagawa, S., Maier, L., Rakova, N., Schatz, V., Neubert, P., … Müller, D. N. (2017). Salt-responsive gut commensal modulates TH17 axis and disease. Nature, 551(7682), 585–589. https://doi.org/10.1038/nature24628



