You are much more than just you. As you made your way into the world, you began a lifelong relationship with microbes. Exactly where, when, and how this relationship began is a topic of ongoing research that sheds light on exactly where we fit in a much larger picture.

The term “microbiome” refers to a community of microorganisms, their components (like nucleic acids and metabolites), and the surrounding environmental conditions. In the case of your own personal microbiome, you’re home to trillions of microorganisms living on and inside you, including an estimated 38,000,000,000,000 bacteria.1 Most live in your gastrointestinal tract, while others live in areas like your mouth, on your skin, in your belly button, and under your armpits.
Scientists now know our gut microbiome plays a central role in many, if not all, aspects of our health and everyday functioning including digestion, metabolism, immunity, skin health, cognition, and more. It even influences some of our behaviors and moods. In states of disorder or disease, there can often be an associated imbalance or irregularity in the microbiota.
So, where does the microbiome come from? And how does it develop?
As a baby transitions from living and growing inside their carrier, to birth, another biological miracle occurs: the development of the microbiome and the beginning of a lifelong relationship with microbes—a relationship that is in constant flux.
The first microbes you are exposed to are your biological mother’s, through a process called “seeding” (ever wondered why we’re called Seed? 😉) that begins in the womb, crescendos at birth with an introduction to the outside world at delivery, and then continues through early infancy and physical development in early childhood. There are important gut microbiome developments thereafter, influenced by factors like nutrition, household environmental exposures, family contacts, pets, and the use of antibiotics.
Here, we’ll mainly focus on the development from birth to 2–3 years of age, at which point the gut microbiota begin to “stabilize” into what is known as a “steady state” microbiome, resembling more like that of an adult.
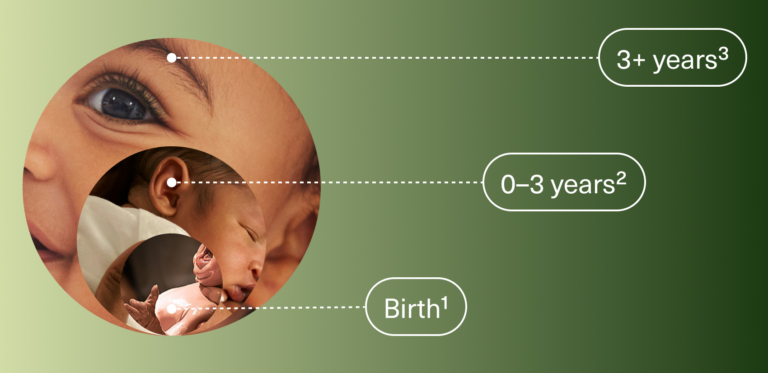
1 0 months/birth: Seeding begins
2 0-3 years: The microbiome is rapidly changing and evolving
3 3+ years: The gut microbiome stabilizes into what is known as a “steady state” microbiome
Definitions
Words and phrases to understand when learning about how the infant microbiome develops:
Definitions
Microbe: Any sort of microscopic organism, or microorganism, including bacteria, archaea, protists, fungi, and viruses.
Microbiota: The microorganisms living in a specific environment (such as your body or a bee) or on your body.
Microbiome: The cells, components, and genomes of all the microbes in a particular environment, for example, in or on a human.
Components: The spectrum of molecules produced by a microorganism, including nucleic acids, metabolites, and other molecules produced by coexisting hosts.
Prenatal Period: The period of time before birth.
Perinatal Period: The period of time immediately before and after birth.
Neonatal Period: The first four weeks of a child’s life after birth.
Seeding: The first exposure to the mother’s microbes at birth.
Weaning: The process of transitioning a baby off of breastfeeding.
Steady State: The adult composition of the gut microbiome, which begins to stabilize between the ages of 3–5. The dominant species in an adult microbiome tend to remain relatively unchanged and “steady” from this stage forward, although the composition can shift slightly under the influence of outside factors such as diet, exercise, medicine, and sleep.
Are babies born with a microbiome?
Up until recently, the answer to this question has been no. But, while the general consensus has long been that the prenatal environment is “sterile”2, recent research has led some scientists to believe that the seeding of the microbiome begins before birth. For example, several researchers have presented evidence of bacteria in the placenta3 (the organ that connects the mother to the baby), meconium4 (the substance that lines the baby’s intestines during pregnancy), and amniotic fluid5 (the fluid in the amniotic sac that contains and protects a fetus in the womb).
However, other scientists argue that it’s not enough to show evidence of bacteria in the in utero environment.6 They want proof that there is an established microbial community before asserting babies have a microbiome at birth. At this time, it’s not yet clear whether those bacteria are simply passing through the fetus, or whether they’re actually growing, dividing, and living in the fetal gut.
More research is needed to validate the presence of a functional fetal microbiome, to find out if or how the microbes are accumulating in the fetus, to explore the role they may play in fetal development, and to explain how they are participating in the education of the fetal immune system to better meet the outside world. To a great extent, however, the argument over the exact presence and extent of a fetal microbiome is still open to debate. But what we do know: the developing fetus is bathed throughout gestation by the metabolic products that stream from the mother’s own microbiome, priming the fetus for independent life and helping to build its metabolism and immune system.
Birth is the major event when we know that newborns are “seeded” with their biological mothers’ microbiota.7 This initial microbial exposure builds the foundation for the developing microbiome8,9, and these early microbes play a key role in everything from metabolism and immune system function, to the digestive system and brain development.
The question of where these initial microbes come from, however, still remains. Scientists have thought in the past that newborns were acquiring mainly vaginal bacteria, but another study assessed infants born via cesarean and vaginal birth, and found that the majority of an infant’s gut microbiome can be traced to their mother’s gut microbiota, regardless of delivery route.10
The same study also found that infants born via cesarean had gut microbiomes with a different composition than those born vaginally. Interestingly, researchers have found that by the time the infants were weaned (at the age of 6–9 months) the differences in the gut microbiomes of babies delivered vaginally versus by cesarean had mostly disappeared.11
Questions remain about the long-term implications of this period when microbes differ depending on birth mode, because the first weeks of life are important for the development of a baby’s immune system.12 That said, birth mode, while important, is not the only factor that contributes to the developing gut microbiome during this time, and there are many ways to foster a resilient microbial community in young children.
Another factor that seems to play a role in the development of a child’s gut microbiome and, in particular, the gut flora during this period, includes the use of antibiotics, which are given to approximately half of women during labor and delivery in the United States.13 During a typical cesarean birth, for example, mothers are given intrapartum antibiotic prophylaxis (IAP), and while the exact implications of antibiotics aren’t known, one important clue about their impact is their association with decreased diversity of several types of bacteria in the infant gut that are associated with higher incidences of childhood allergies.
After birth and before the introduction of solid food, an infant’s gut microbiome is turbulent, changes quickly, and is influenced by a variety of factors, such as nutrition and breastfeeding.
Breastfeeding influences the overall health of the baby for a variety of reasons, including the development of the gut microbiome. There are various components of breastfeeding that may impact an infant’s microbiome:
- Human milk oligosaccharides (HMOs): HMOs are a prominent component of breast milk, which aren’t digested but serve as a prebiotic for beneficial Bifidobacterium. While the research isn’t yet definitive, scientists think that an abundance of Bifidobacterium in the infant gut is important because it may result in lower levels of potential gut pathogens9, reduced intestinal inflammation14, and fewer antibiotic resistant microbial genes.15
- Bacteria in breast milk: While breast milk was once considered a sterile fluid, we now know that human breast milk contains an abundance of bacterial species.16 These bacteria are thought to enhance infant health by preventing pathogen adhesion and promoting gut colonization of beneficial microbes.17 However, the origin of bacteria in breast milk is highly debated, and there are multiple studies underway investigating how exactly bacteria gets into breast milk.18,19 One interesting area being investigated involves the combination of breastmilk and baby’s saliva. Studies have shown that bacteria from the baby’s mouth may move back into the mammary gland and influence the types and quantities of bacteria passed via milk.20 At the same time, breastmilk and saliva interactions may support immunity by acting together to regulate the oral microbiome of newborn babies.21
- Skin-to-skin contact: The skin-to-skin contact occurring during breastfeeding may also influence the developing microbiome, as distinct bacterial communities of the areola have been established. In fact, a team of scientists at UCLA found that 10% of the beneficial bacteria in a baby’s intestinal tract came from the skin on the mother’s breast.22
Research has demonstrated differences in the intestinal microbiome and growth outcomes between exclusively breast versus formula-fed infants (though very little is known about the effects of introducing formula to breastfed infants, either briefly or long term, on these outcomes).While there is a strong correlation between breastfeeding and positive health outcomes for a child, this may not be feasible for many mothers. For caretakers who partake in formula feeding, the good news is that the infant formula industry is evolving by striving to design infant formulas that more closely “mimic” breast milk through the addition of HMOs and other constituents absent from or under-abundant in formula.23
The final large shift in the infant microbiome happens with the transition from breastmilk or formula to solid foods. At this point, the gut microbiome is becoming more stable and has begun to closely resemble that of an adult, with, among other things, fewer Bifidobacterium and more bacteria from the Bacteroidetes and Firmicutes phyla.24 As mentioned above, this is known as a “steady state” microbiome.
While nutrition is central to the development of the infant’s microbiome, several other factors can play a role, including pets, household composition, antibiotics, outdoor exposures, exercise, and early life adversity. Here’s what a few studies show:
Pets: Exposure to animals informs the gut microbiome of infants, according to some studies.25 In one study, children exposed to pets in early life experienced significant increases in two beneficial gut bacteria: Ruminococcus and Oscillospira.26 Further, clinical studies have concluded that early exposure to household pets is associated with a significant decrease in childhood and adult allergies, asthma, and even obesity.
Household: While data is mixed, there is some evidence that the social environment of infants (represented by variations in household composition, and illustrated through an activity like crawling on the floor) may influence the bacterial composition of the infant gastrointestinal microbiome. It is thought that a higher number and diversity of cohabitants and potential caregivers may facilitate social transmission of beneficial bacteria to the infant gastrointestinal tract, by way of shared environment or through direct physical and social contact.27
Antibiotics: As mentioned previously, use of antibiotics in early life has been correlated to a variety of diseases and conditions, particularly allergies.28 While scientists don’t yet know how, it seems that antibiotics delay the natural maturation of the child gut microbiome.29
Early Life Adversity: Research in animals suggests that early life stress leads to decreased microbial diversity, which links to poorer health outcomes.30 In another study, a subset of persons with IBS were more likely to have experienced early life trauma, and displayed a gut microbial composition that was different from a healthy control group.31
Antimicrobials: As we know, sanitization is imperative for combating disease and community spread. But greater emphasis on cleanliness in the last century has led to widening use of antimicrobials, including disinfectants and other cleaning agents in the home. These products work by killing microorganisms, or stopping their growth—but may also disrupt the microbiome. One study looked at associations between household cleaning product use and infant gut microbial composition at 3–4 months of age, finding that exposure to household disinfectants was associated with higher body mass index (BMI) at age 3.32
While there are many factors that impact the microbiome within the first few years of life (birth mode, feeding, antibiotics), keep in mind that these are not the end-all, be-all for determining the composition of the microbiome.
Even after reaching a “steady state”, the composition of the microbiome continues to undergo shifts throughout life. This means that while the composition (what microbes are present, in what proportions) remains relatively stable, this composition can continue to shift, shaped by each individuals’ life experience, interactions, environment, and daily choices.33
Because there are a myriad of factors that can shape each person’s assemblage of microbial life, every individual’s microbiome is completely unique to them, like a living fingerprint. No two people (not even twins) share the same microbial makeup. In this way, there is no fixed state, no universal endpoint, no one “perfect” microbiome. Instead, the microbiome is continually evolving with us throughout life, influenced by daily actions, behaviors and practices.
—
The views expressed in this article intend to highlight scientific studies and current opinion from the scientific and medical literature. They are the views of the author and are for informational purposes only, even if and to the extent that this article features the advice of physicians and medical practitioners. This article is not, nor is it intended to be, a substitute for professional medical advice, diagnosis, or treatment, and should never be relied upon for specific medical advice.
- Sender R, Fuchs S, Milo R (2016) Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol 14(8): e1002533. https://doi.org/10.1371/journal.pbio.1002533
- Blaser, M.J., Devkota, S., McCoy, K.D. et al. Lessons learned from the prenatal microbiome controversy. Microbiome 9, 8 (2021). https://doi.org/10.1186/s40168-020-00946-2
- Aagaard K, Ma J, Antony KM, Ganu R, Petrosino J, Versalovic J. The placenta harbors a unique microbiome. Sci Transl Med. 2014 May 21;6(237):237ra65. doi: 10.1126/scitranslmed.3008599. PMID: 24848255; PMCID: PMC4929217.
- Ardissone AN, de la Cruz DM, Davis-Richardson AG, Rechcigl KT, Li N, Drew JC, Murgas-Torrazza R, Sharma R, Hudak ML, Triplett EW, Neu J. Meconium microbiome analysis identifies bacteria correlated with premature birth. PLoS One. 2014 Mar 10;9(3):e90784. doi: 10.1371/journal.pone.0090784. Erratum in: PLoS One. 2014;9(6):e101399. PMID: 24614698; PMCID: PMC3948723.
- Collado MC, Rautava S, Aakko J, Isolauri E, Salminen S. Human gut colonisation may be initiated in utero by distinct microbial communities in the placenta and amniotic fluid. Sci Rep. 2016 Mar 22;6:23129. doi: 10.1038/srep23129. PMID: 27001291; PMCID: PMC4802384.
- Briana DD, Papaevangelou V, Malamitsi-Puchner A. The jury is still out on the existence of a placental microbiome. Acta Paediatr. 2021 Nov;110(11):2958-2963. doi: 10.1111/apa.16048. Epub 2021 Jul 31. PMID: 34319607.
- Dunn, A. B., Jordan, S., Baker, B. J., & Carlson, N. S. (2017). The Maternal Infant Microbiome: Considerations for Labor and Birth. MCN. The American journal of maternal child nursing, 42(6), 318–325. https://doi.org/10.1097/NMC.0000000000000373
- Ferretti, P., Pasolli, E., Tett, A., Asnicar, F., Gorfer, V., Fedi, S., Armanini, F., Truong, D. T., Manara, S., Zolfo, M., Beghini, F., Bertorelli, R., de Sanctis, V., Bariletti, I., Canto, R., Clementi, R., Cologna, M., Crifò, T., Cusumano, G., . . . Segata, N. (2018). Mother-to-Infant Microbial Transmission from Different Body Sites Shapes the Developing Infant Gut Microbiome. Cell Host & Microbe, 24(1), 133–145.e5. https://doi.org/10.1016/j.chom.2018.06.005
- Smilowitz, J. T., & Taft, D. H. (2020b, June 1). The Infant Gut Microbiome and Probiotics that Work. The Scientist Magazine®. https://www.the-scientist.com/features/the-infant-gut-microbiome-and-probiotics-that-work-67563
- Maqsood, R., Rodgers, R., Rodriguez, C., Handley, S. A., Ndao, I. M., Tarr, P. I., Warner, B. B., Lim, E. S., & Holtz, L. R. (2019). Discordant transmission of bacteria and viruses from mothers to babies at birth. Microbiome, 7(1). https://doi.org/10.1186/s40168-019-0766-7
- Shao, Y., Forster, S. C., Tsaliki, E., Vervier, K., Strang, A., Simpson, N., Kumar, N., Stares, M. D., Rodger, A., Brocklehurst, P., Field, N., & Lawley, T. D. (2019). Stunted microbiota and opportunistic pathogen colonization in caesarean-section birth. Nature, 574(7776), 117–121. https://doi.org/10.1038/s41586-019-1560-1
- Neu, J., & Rushing, J. (2011). Cesarean versus vaginal delivery: long-term infant outcomes and the hygiene hypothesis. Clinics in perinatology, 38(2), 321–331. https://doi.org/10.1016/j.clp.2011.03.008
- Moore, R. E., & Townsend, S. D. (2019). Temporal development of the infant gut microbiome. Open Biology, 9(9), 190128. https://doi.org/10.1098/rsob.190128
- Henrick, B.M., Chew, S., Casaburi, G. et al. Colonization by B. infantis EVC001 modulates enteric inflammation in exclusively breastfed infants. Pediatr Res 86, 749–757 (2019). https://doi.org/10.1038/s41390-019-0533-2
- Casaburi, G., Duar, R.M., Vance, D.P. et al. Early-life gut microbiome modulation reduces the abundance of antibiotic-resistant bacteria. Antimicrob Resist Infect Control 8, 131 (2019). https://doi.org/10.1186/s13756-019-0583-6
- Fernández L, Langa S, Martín V, Maldonado A, Jiménez E, Martín R, Rodríguez JM. The human milk microbiota: origin and potential roles in health and disease. Pharmacol Res. 2013 Mar;69(1):1-10. doi: 10.1016/j.phrs.2012.09.001. Epub 2012 Sep 10. PMID: 22974824.
- Lyons, K. E., Ryan, C. A., Dempsey, E. M., Ross, R. P., & Stanton, C. (2020). Breast Milk, a Source of Beneficial Microbes and Associated Benefits for Infant Health. Nutrients, 12(4), 1039. https://doi.org/10.3390/nu12041039
- Rodríguez J. M. (2014). The origin of human milk bacteria: is there a bacterial entero-mammary pathway during late pregnancy and lactation?. Advances in nutrition (Bethesda, Md.), 5(6), 779–784. https://doi.org/10.3945/an.114.007229
- Jeurink PV, van Bergenhenegouwen J, Jiménez E, Knippels LM, Fernández L, Garssen J, Knol J, Rodríguez JM, Martín R. Human milk: a source of more life than we imagine. Benef Microbes. 2013 Mar 1;4(1):17-30. doi: 10.3920/BM2012.0040. PMID: 23271066.
- Moossavi, S., Sepehri, S., Robertson, B., Bode, L., Goruk, S., Field, C. J., Lix, L. M., de Souza, R. J., Becker, A. B., Mandhane, P. J., Turvey, S. E., Subbarao, P., Moraes, T. J., Lefebvre, D. L., Sears, M. R., Khafipour, E., & Azad, M. B. (2019). Composition and Variation of the Human Milk Microbiota Are Influenced by Maternal and Early-Life Factors. Cell Host & Microbe, 25(2), 324–335.e4. https://doi.org/10.1016/j.chom.2019.01.011
- Al-Shehri, S. S., Knox, C. L., Liley, H. G., Cowley, D. M., Wright, J. R., Henman, M. G., Hewavitharana, A. K., Charles, B. G., Shaw, P. N., Sweeney, E. L., & Duley, J. A. (2015). Breastmilk-Saliva Interactions Boost Innate Immunity by Regulating the Oral Microbiome in Early Infancy. PLOS ONE, 10(9), e0135047. https://doi.org/10.1371/journal.pone.0135047
- Pannaraj, P. S., Li, F., Cerini, C., Bender, J. M., Yang, S., Rollie, A., Adisetiyo, H., Zabih, S., Lincez, P. J., Bittinger, K., Bailey, A., Bushman, F. D., Sleasman, J. W., & Aldrovandi, G. M. (2017). Association Between Breast Milk Bacterial Communities and Establishment and Development of the Infant Gut Microbiome. JAMA Pediatrics, 171(7), 647. https://doi.org/10.1001/jamapediatrics.2017.0378
- Notman, N. (2021, November 29). The science of breast milk and baby formula. Chemistry World. https://www.chemistryworld.com/features/the-science-of-breast-milk-and-baby-formula/4014574.article
- Dong, T. S., & Gupta, A. (2019). Influence of Early Life, Diet, and the Environment on the Microbiome. Clinical Gastroenterology and Hepatology, 17(2), 231–242. https://doi.org/10.1016/j.cgh.2018.08.067
- Azad, M. B., Konya, T., Maughan, H., Guttman, D. S., Field, C. J., Sears, M. R., Becker, A. B., Scott, J. A., & Kozyrskyj, A. L. (2013). Infant gut microbiota and the hygiene hypothesis of allergic disease: impact of household pets and siblings on microbiota composition and diversity. Allergy, Asthma & Clinical Immunology, 9(1). https://doi.org/10.1186/1710-1492-9-15
- Tun, H.M., Konya, T., Takaro, T.K. et al. Exposure to household furry pets influences the gut microbiota of infants at 3–4 months following various birth scenarios. Microbiome 5, 40 (2017). https://doi.org/10.1186/s40168-017-0254-x
- Lane AA, McGuire MK, McGuire MA, Williams JE, Lackey KA, Hagen EH, Kaul A, Gindola D, Gebeyehu D, Flores KE, Foster JA, Sellen DW, Kamau-Mbuthia EW, Kamundia EW, Mbugua S, Moore SE, Prentice AM, Kvist LJ, Otoo GE, Rodríguez JM, Ruiz L, Pareja RG, Bode L, Price WJ, Meehan CL. 2019. Household composition and the infant fecal microbiome: the INSPIRE study. Am J Phys Anthropol 169:526–514.
- Ahmadizar, F., Vijverberg, S. J. H., Arets, H. G. M., de Boer, A., Lang, J. E., Garssen, J., Kraneveld, A., & Maitland-van Der Zee, A. H. (2017). Early-life antibiotic exposure increases the risk of developing allergic symptoms later in life: A meta-analysis. Allergy, 73(5), 971–986. https://doi.org/10.1111/all.13332
- Bokulich, N. A., Chung, J., Battaglia, T., Henderson, N., Jay, M., Li, H., D. Lieber, A., Wu, F., Perez-Perez, G. I., Chen, Y., Schweizer, W., Zheng, X., Contreras, M., Dominguez-Bello, M. G., & Blaser, M. J. (2016). Antibiotics, birth mode, and diet shape microbiome maturation during early life. Science Translational Medicine, 8(343). https://doi.org/10.1126/scitranslmed.aad7121
- Moussaoui, N., Jacobs, J. P., Larauche, M., Biraud, M., Million, M., Mayer, E., & Taché, Y. (2017). Chronic Early-life Stress in Rat Pups Alters Basal Corticosterone, Intestinal Permeability, and Fecal Microbiota at Weaning: Influence of Sex. Journal of Neurogastroenterology and Motility, 23(1), 135–143. https://doi.org/10.5056/jnm16105
- Labus, J.S., Hollister, E.B., Jacobs, J. et al. Differences in gut microbial composition correlate with regional brain volumes in irritable bowel syndrome. Microbiome 5, 49 (2017). https://doi.org/10.1186/s40168-017-0260-z
- Tun, M. H., Tun, H. M., Mahoney, J. J., Konya, T. B., Guttman, D. S., Becker, A. B., Mandhane, P. J., Turvey, S. E., Subbarao, P., Sears, M. R., Brook, J. R., Lou, W., Takarao, T. K., Scott, J. A., & Kozyrskyj, A. L. (2018). Postnatal exposure to household disinfectants, infant gut microbiota and subsequent risk of overweight in children. Canadian Medical Association Journal, 190(37), E1097–E1107. https://doi.org/10.1503/cmaj.170809
- Voreades, N., Kozil, A., & Weir, T. L. (2014). Diet and the development of the human intestinal microbiome. Frontiers in Microbiology, 5. https://doi.org/10.3389/fmicb.2014.00494



